
There are a couple of reasons why I’m going to discuss the comedy of errors surrounding my identification of this patch of Hypnum subimponens. For one thing I think it could be instructional for would-be naturalists of all stripes, since it illustrates many of the possible pitfalls inherent in the identification process, many of which have nothing to do with lab techniques, microscopy, or getting a solid handle on terminology. And secondly I am hoping that publicly excoriating myself will drive these lessons home, and that that embarrassment will trigger a red flag the next time I’m inclined to be shoddy in any of these phases of the process.

I found this patch of moss on my initial trip to The Cliff, and my first mistake was in thinking it was Kindbergia praelonga, based on…well, based on the fact that I frequently think I know things which I do not, in fact, know. And because I thought I knew what it was I didn’t bother to label the bag with date, location, or photo number (since for the purposes of this blog an identification without a photo is useless). Disorganized fool that I often can be, I did the same thing with a new specimen a few weeks later. One would think that by now I would know that my memory is about as reliable as a poorly maintained, $500 rustbucket, and for similar reasons.

A couple weeks after that I made another trip to The Cliff, bagged and tagged the specimen appropriately, rinsed it when I got home and placed it in a wet dish with identifying information on it. And then promptly forgot about it because I still believed I knew what it was, and I had more ‘interesting’ lichens and mosses to work on. When I got back to it several days later I found that, because I had left it with too much water and inadequate ventilation, it had molded.

The next time I made it to The Cliff I grabbed a small sample, labeled it with every bit of info I could think of, and began the identification process. Where I soon realized that this was definitely not Kindbergia praelonga. So I started at the beginning of the Brachytheciaceae family key, but it was all square pegs and round holes. Eventually I stumbled into the idea that it might be a Hypnum spp., which Terry McIntosh confirmed, and then asked me what the stem cross section looked like. Except that, of course, I had not paid any attention to what I did with those stems after I stripped the leaves, and could not provide a cross section.

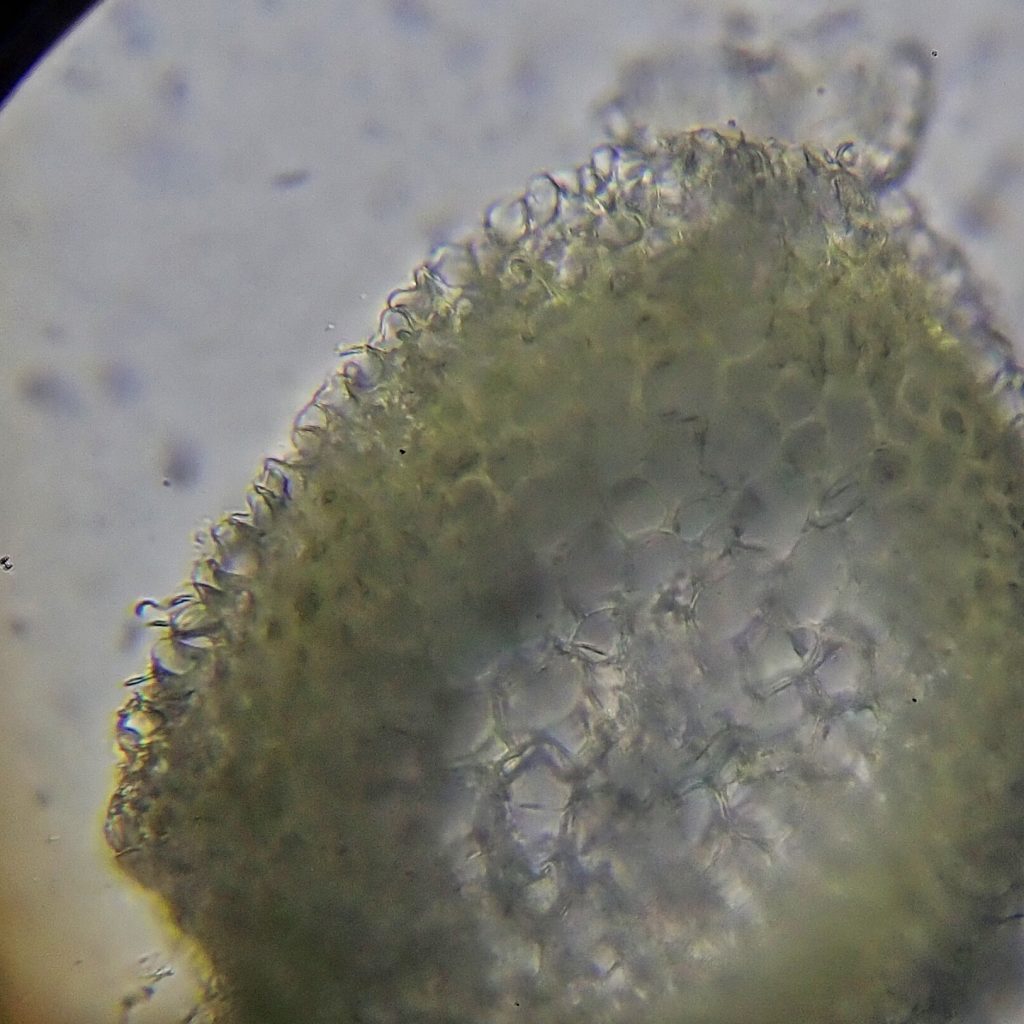
Stem cross sections are very important in the identification of Hypnum spp. That is because the first couplet in every key to them starts with presence/absence of hyalodermis. There are over a dozen species of Hypnum in our region, and they are split roughly in half by this couplet. A hyalodermis is a set of enlarged, clear (hyaline), thin walled cells that form an outer layer (dermis) around the stem. They are used for water storage, and they can only be seen under high magnification (these photos are 400x).
I was very frustrated with myself, and didn’t go out to The Cliff for almost a month after that debacle. But I will say that everything I collected during that month was well labeled with date, location and photo number, and I was able to easily match identification to specimen photos. But, unfortunately, I hadn’t extended my new found rigorous organization to the lab bench, because the next time I tried to identify something from that patch I somehow contaminated it with pieces from another species of moss, which thoroughly confused the matter.

And there was another, more technical issue. Because of my relative clumsiness I chose to cut cross sections from the biggest, thickest, stem I could find. And, although I had to cut dozens of them to achieve it, I finally sliced a couple of them that were thin enough to lay flat on a slide. Because they lacked a hyalodermis I went that way in the key and ended up at a subspecies of H. revolutum which has less revolute leaf margins. I sent photos off to Terry McIntosh, thinking I was done.

But I wasn’t. Terry said that the leaves didn’t quite look right to him for H. revolutum, and that possibly the hyalodermis had degraded, since there appeared to be shreds of larger cells. He suggested I try a cross section of a younger shoot, or further towards the tip of the one I had. So, since my clump was old and dried, I made another trip to The Cliff yesterday, promising myself I would only grab some of the Hypnum, and a sprig of the Cladonia furcata I wrote about yesterday.

This time I was very methodical at my lab bench. I took a single shoot with a seta and immature capsule, rinsed it thoroughly in dechlorinated water, and photographed it under the stereoscope. Next I removed the branches and made a slide of them. Then I carefully stripped the stem leaves and made a slide of them. And finally I put the stem under the stereoscope and diced a full inch of it into the thinnest slices I could manage. Then I added a couple drops of water to the slices and manipulated them under the stereoscope until some of them seemed to be lying flat.
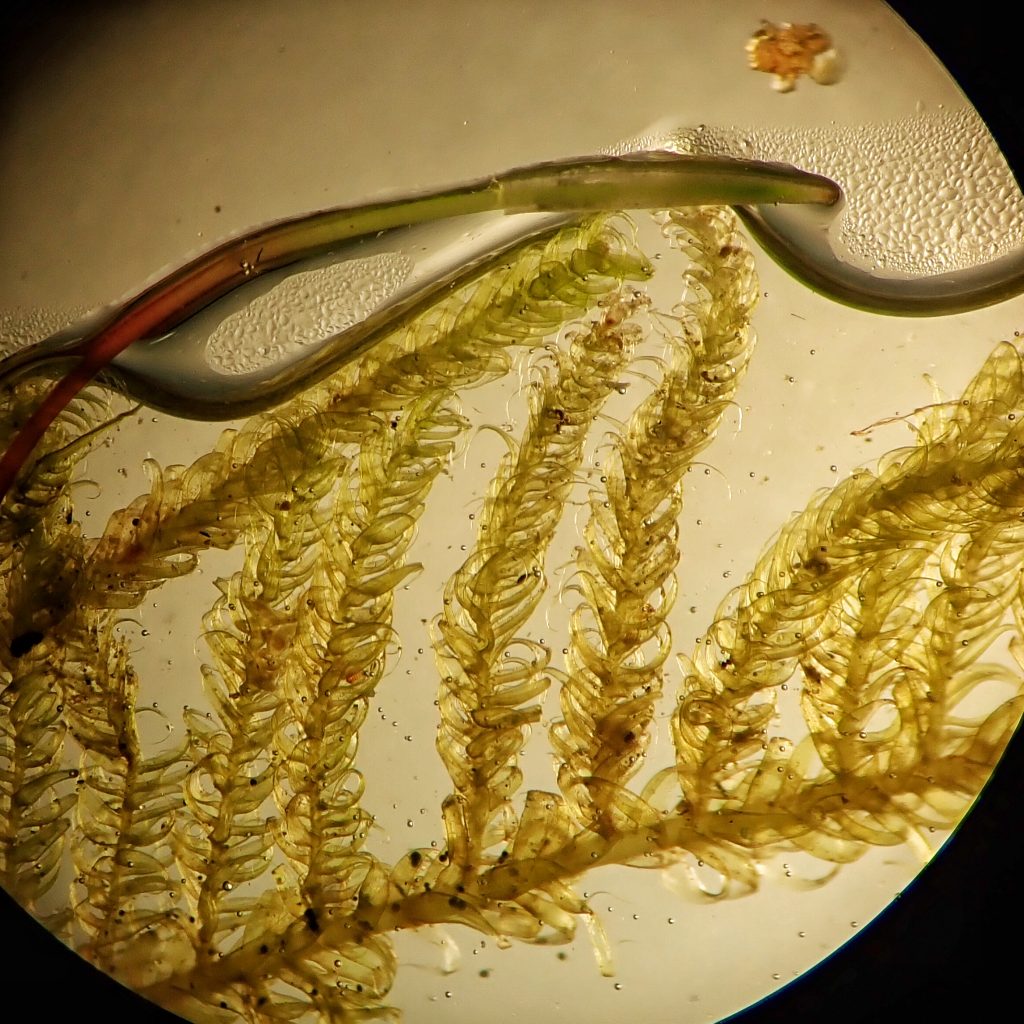
I slapped a cover slip on that slide, slid it under the compound microscope, and, after some spatial and magnification adjustments, there it was- a hyalodermis! I checked as many different cross sections as I could and it was clearly there in all of them. Then back to the leaf slide. Alar cells quadrate and not inflated. Leaves gradually tapering to insertion. Leaves circinate. Leaves over 1.5 mm. Hypnum subimponens! And Terry McIntosh confirmed it, probably almost as glad to see the end of that chapter as I was.

So, this was pretty long winded, and probably boring to many. I’d guess I lost half my readers several paragraphs ago. But for those of you who made it through I hope I can be a good bad example about the complications, and general bad science, of not being methodical and organized in the processing of specimens. Besides wasting a lot of time I also squandered the lives of those mosses. It is not a critical loss, and I won’t lose sleep over it, but I’d feel better if I hadn’t. I’d like to think I have finally learned that lesson well, but only time will tell.
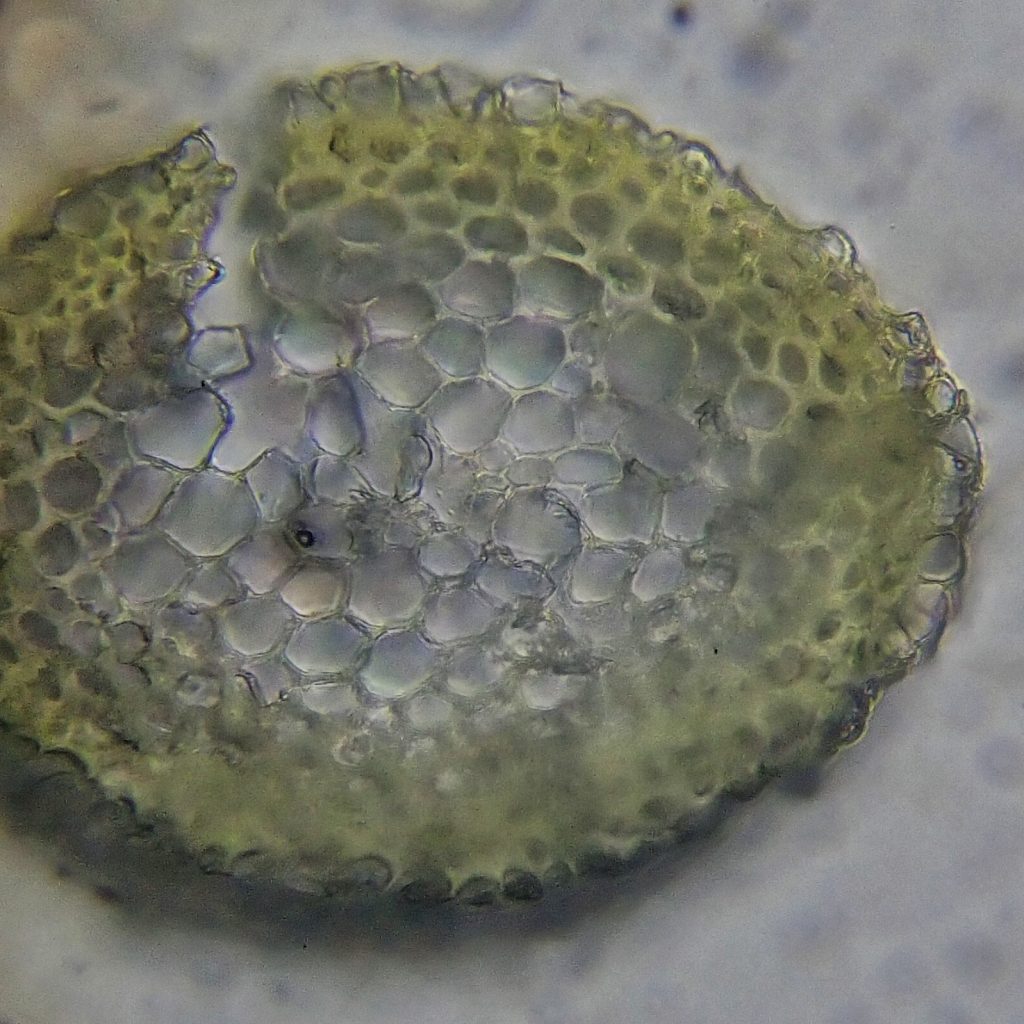
Description-Yellow-green, prostrate and pleurocarpous moss that forms dense mats; branching is regularly pinnate, sometimes dense enough to be feather-like; stems have a hyalodermis; alar cells are not inflated and are often rectangular; leaves are ovate, wide near the base and gradually narrowing to a long apex, which curls back into the leaf and almost forms a circle (circinate); costa is short and doubled, and the leaves gradually narrow to the base; capsule smooth, cylindrical, and slightly curved.

Similar species– Educated guesses can be made, but it is impossible to positively identify Hypnum spp. without extensive experience or a key, and lab techniques and microscopy, due to the importance of ascertaining the presence or absence of the hyalodermis; H. circinale lacks a hyalodermis, is whitish green, forms braided looking mats, and has narrower leaves and narrower shoots; H. pratense leaves are quite rounded to their attachment, alar cells differentiated and often inflated, and it is rare in the PNW; H. lindbergii has clearly inflated alar cells, falcate (hooked at the apex) rather than circinate leaves, and the capsule is ribbed when dry; H. revolutum lacks a hyalodermis, and its leaf margins are revolute at least to the middle of the leaf.

Habitat-Epiphytic on trees, logs, and rocks in wet to mesic forests up to 4,000’.
Range– Northwestern North America, eastern Asia; most common in our region from the the east slope of the Cascades west to the Pacific Ocean; also found in the montane regions of ne Washington, ne Oregon, n Idaho, nw Montana.
Reproductive timing– Early spring to early summer.

Etymology of names–Hypnum refers to the Greek word for sleep. This is because members of this genus were used to stuff pillows, and this genus was possibly used for that because of an oracular pronouncement that they possessed a magic to confer happy dreams. The specific epithet subimponens means that it is similar in variability of appearance to H. imponens, from the Latin word for imposter, because that moss sometimes bears a strong resemblance to other Hypnum spp.

http://www.efloras.org/florataxon.aspx?flora_id=1&taxon_id=250064913
http://linnet.geog.ubc.ca/Atlas/Atlas.aspx?sciname=Hypnum%20subimponens
http://fieldguide.mt.gov/speciesDetail.aspx?elcode=NBMUS3V0J0
https://wildflowersearch.org/search?&PlantName=Hypnum+subimponens

Yep, it’s called pulling a Craig. In spite of overwhelming evidence, I still put trust in my memory and first impressions. 🤦♂️
🤣😂As someone else commented on Facebook, from their experience, at least my research site wasn’t far into the backcountry and accessible only by boat
Fantastic Post! I love the stories of doing what naturalists do. I am excited to investigate the mosses of Alaska (once the 5′ of snow melts) and your post inspired me to print out this: http://flora.huh.harvard.edu/FloraData/001/WebFiles/fna27/InsertJPG/1Morph1.jpg. so I could determine what the heck you are talking about.
I totally identify with “thinking I know things” that I may not know. Mine are much simpler things but same deal.
Carry on, being human and telling us so we can imagine the process of ID-ing as possible. Do you mind if I ask what exact tool you are using for the magnified photos?
Thank you for your kind words. I have an Olympus TG5 that I place over the ocular of either the stereoscope or the compound microscope. It’s also what I use for macro photography in the field, because it has built in photo stacking.
Nice graphics on that link. I also post a link to the Flora of North America page for each moss I profile. It’s usually the top link. Good luck with your mosses!
BTW buddy, I’ll gladly surrender the coinage to you! 🤣
🤣😂
Around where I live (dryer areas of southern Vancouver Island), I mostly find Hypnum subimponens and H. circinale. When I spot the moss on the top of damp logs and it has rounded, blunted sporangia, it’s usually H. circinale (perhaps “circinale” a reference to the almost-round sporangia?)
You may be correct, but I thought circinale referrred to the circinate leaves. Thanks for the info on the capsules. I’ll look for that.